Cruxi Add to favorites
Last update time : 2025-12-17 12:31:15
Cruxi automates FDA and global medical device submissions using AI. Eliminate Refusal-to-Accept risks and accelerate market entry with expert-verified dossiers.
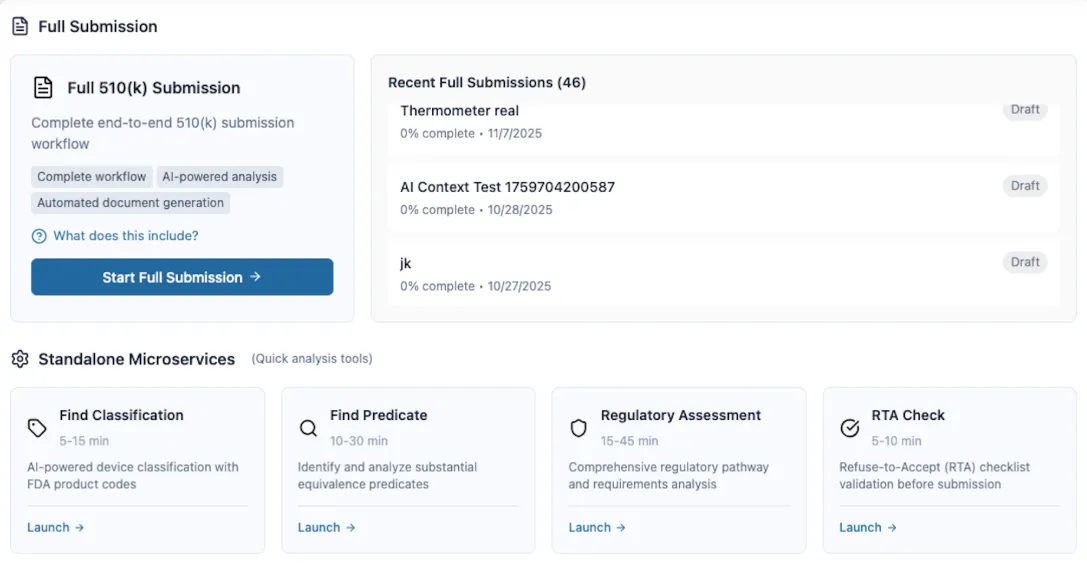
Cruxi is revolutionizing the medical device landscape with the launch of its sophisticated AI-powered regulatory submission platform. Designed specifically for medtech teams, the platform automates the most labor-intensive aspects of global filings, including FDA 510(k), De Novo, PMA, and IDE submissions. By integrating AI-driven device classification with intelligent predicate finding, Cruxi simplifies the pathway from development to market approval.
The platform’s core strength lies in its ability to draft eSTAR documents and perform automated compliance checks, significantly reducing 'Refusal-to-Accept' (RTA) risks. Unlike generic AI tools, Cruxi’s models are trained exclusively on official FDA guidance and extensive regulatory databases. This ensures that every generated output includes traceable citations, supporting the company's commitment to 'zero-hallucination' accuracy. To maintain the highest standards of safety and compliance, the system incorporates a human-in-the-loop review process, allowing regulatory experts to verify AI-generated content before submission.
Security and reliability remain central to the Cruxi experience. The platform features enterprise-grade security protocols and receives continuous regulatory updates to keep pace with evolving international standards. By automating evidence planning and package exports, Cruxi enables medical device manufacturers to produce reviewer-ready, high-quality dossiers in a fraction of the traditional time, ultimately helping life-saving technologies reach patients faster.
Pricing : Paid
Web Address : Cruxi
Tags : Cruxi medical device regulatory software FDA 510k automation AI medtech eSTAR drafting medical device compliance PMA submission tool regulatory submission platform
Similar AI tools
TradeUI
Bizora
WinDispute.pro
Kick
Zola Analytics
BeeBee Ai
Payroll Robot
RAFA
Candlestick
MLQ App
GetInvoice
DealWorthIt
AI Tools
- Aggregators
- AI Detection
- Automation & Agents
- Avatar Creators
- Chatbots
- Copywriting
- Finance
- For fun
- Games
- Generative Art
- Generative Code
- Generative Video
- Image Improvement
- Inspiration
- Marketing
- Motion Capture
- Music
- Personal Development
- Podcast
- Productivity
- Prompt Guides
- Research
- Social Media
- Speech to Text
- Text to Speech
- Text to Video
- Translation
- Video Editing
- Visual Scanning & Analysis
- Voice Modulation